2. Müller Glial Cell Reactive Gliosis and Adult Retinal Regeneration
In contrast to mammals, zebrafish possess the remarkable ability to regenerate retinal neurons. Damage to the zebrafish retina induces Müller glia into a stem cell fate, generating retinal progenitors for regeneration. In contrast, injury in the mammal retina results in Müller glial reactive gliosis, a characteristic gliotic response that is detrimental to vision. Understanding the signaling pathways that determine how Müller glial cells respond to injury is a critical step toward promoting regeneration in the mammalian retina. We found that cell cycle inhibition forced zebrafish Müller glia to become persistently reactive, which resulted in a complete absence of regeneration. Conversely, overexpression of Sonic Hedgehog (Shh), a key signaling molecule during retinal development, resulted in unregulated Müller glial hyperproliferation and improper regeneration. These data demonstrate that zebrafish Müller glia possess both gliotic and regenerative potential and characterize a role for Shh during retinal regeneration. The two models of reactive gliosis will serve as a useful platform to screen the adult zebrafish retina for signaling pathways that push Müller glial cells towards gliotic or regenerative cell fates.
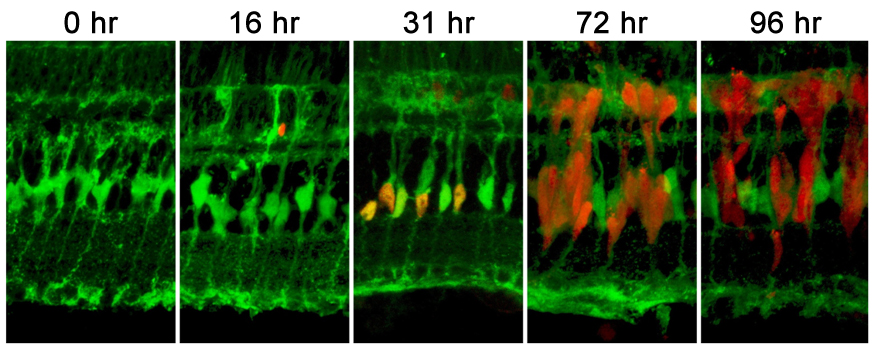
Müller glia are the source of stem cells in the adult zebrafish retina. Retinal sections taken from at Tg(gfap:EGFP) transgenic animal that specifically labels Müller glia with EGFP (green). PCNA expression (red) was used to label proliferating retinal progenitors at various timepoints following damage to the retinal photoreceptors by intense light. This damage event triggers a subset of the Müller glia to express PCNA and re-enter the cell cycle. The resulting progenitor cells continue to proliferate as they migrate to the photoreceptor layer, where they will ultimately replace the lost photoreceptors.